Stark Lab research
Collaborations | Student research projects | Publications
About Dr Romana Stark
My training commenced at the University of Vienna (Austria) where I completed a Masters in Nutrition Science in 2005 and a PhD in Medical Sciences in 2012. My PhD specialised in endocrinology and metabolism with a particular focus on Diabetes. I conducted my PhD research training at Yale University under the guidance of two world leaders in diabetes/obesity research, Prof. Roden (German Diabetes Center, Germany) and Prof. Shulman (Yale University, USA).
In Prof. Shulman's lab, I worked in mitochondrial metabolism and focused on how cells sense energy. I closely worked together with Prof. Shulman's research fellow at this time, A./Prof. Kibbey (Yale University, USA), a pioneer of the 'mitochondrial-GTP (mtGTP) theory' as an energy-sensor and an expert in isotopologue mass spectrometry. I learned to develop novel methodologies to address key research questions. For example, I designed a method to measure mitochondrial substrate flux using mass spectrometry. My research skills do not only include in vivo measurements of complex physiological parameters (e.g. rodent hyperinsulinemic-euglycemic clamps), but also in vitro research in several cell systems and metabolomics using mass spectrometry. My PhD work focused on the role of mitochondrial PEPCK (PEPCK-M) in glucose homeostasis and raised novel aspects about its function, which was highlighted as a key discovery by its inclusion in the Journal of Biological Chemistry Thematic Minireview of PEPCK.
In September 2009 I moved to Australia and joined A./Prof. Watt (Monash University) as a research assistant in the field of lipid metabolism. After I completed my PhD studies in 2012, I joined the lab of Dr Zane Andrews (Monash University, Australia).
Currently, I am a National Health Medical Research Council (NHMRC) Early Career Fellow and investigate neural mechanisms regulating food intake and weight, and how metabolic cues in the brain regulate peripheral tissues in order to maintain energy balance.
Our research
Current projects
- How does the brain sense low glucose levels and switch on corrective actions?
- Brain ghrelin receptor sensing and blood glucose regulation
- The role of ghrelin receptors in the olfactory bulb
- Does mitochondrial PEPCK in the brain acts as energy sensor?
Visit Dr Romana Stark's Monash research profile to see a full listing of current projects.
Research activities
How does the brain sense low glucose levels and switch on corrective actions? We need to understand the mechanisms involved in sensing hypoglycemia to find new avenues for diabetes treatments.
Despite current diabetes management, patients with glucose-lowering therapies often deal with hypoglycemia, a condition with abnormally low blood glucose levels. If hypoglycemia is not corrected, then this can result in cognitive dysfunction including loss of consciousness and seizures; and if prolonged even coma and death. On average, type-1 diabetics suffer from about two mild hypoglycemic episodes per week and have at least one life-threatening severe episode per year. This is also similar for type-2 diabetics requiring insulin therapy. Indeed, insulin-induced hypoglycemia complicates and severely limits the widespread application of insulin, which is for many the only option to treat their diabetes. So how can we help insulin-treated diabetics better manage their blood glucose levels?
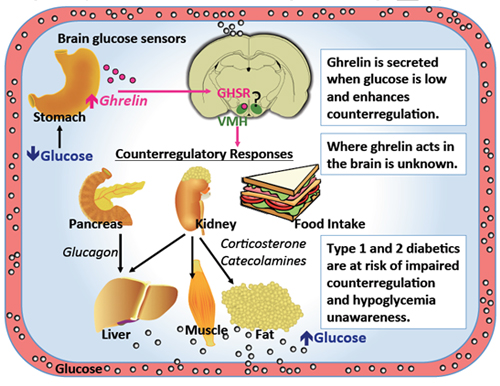
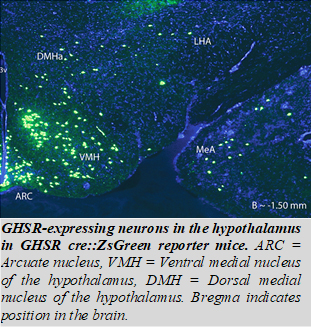
Our lab investigates the neuronal circuits underlying the brain’s ability to sense glucose levels and activate counter-regulation. Research on this topic suggests that the ventromedial nucleus of the hypothalamus (VMH) plays an important role. But how these counter-regulatory responses are coupled with energy deficit, such as during fasting and starvation remains unknown. One key hormone that is secreted from the stomach when energy reserves are low is ghrelin. Our past work shows that ghrelin informs the brain of low energy reserves by binding on ghrelin receptors (GHSR) to stimulate food intake, motivation, and to increase blood glucose. Thus, GHSR-expressing neurons in the brain may couple energy deficit with counter-regulatory response to prevent hypoglycemia. The GHSR neural circuits that regulate blood glucose are unknown but the counter-regulatory role of the VMH and the high expression of GHSR in the VMH, suggesting it is a likely candidate.
Losing one of your senses enhances others. But glucose sensing is not dispensable.
 | 
|

Techniques/expertise
- Metabolic phenotyping
- In-vivo physiology
- Behavioural neuroscience
- Molecular biology
- Optogenetics
- Stereotaxic surgery
Disease models
- Diet-induced obesity
- Type 1 diabetes
Collaborations
We collaborate with many scientists and research organisations around the world. Click on the map to see the details for each of these collaborators (dive into specific publications and outputs by clicking on the dots).
Student research projects
The Stark Lab offers a variety of Honours, Masters and PhD projects for students interested in joining our group. There are also a number of short term research opportunities available.
Please visit Supervisor Connect to explore the projects currently available in our Lab.

