Mifsud Lab research
Collaborations | Student research projects | Publications
About Dr Nicole Mifsud
Dr Mifsud is the Group Leader of the Clinical Immunology Laboratory in the Biomedicine Discovery Institute, Monash University. She was awarded her PhD in Transplantation Immunology from the University of Melbourne in 2005 (supervised by Profs James McCluskey and Brian Tait). She completed postdoctoral training with Prof Weisan Chen (Ludwig Institute for Cancer Research, Melbourne) identifying T cell epitopes in melanoma and influenza and subsequently with A/Prof Tom Kotsimbos (The Alfred Hospital/Monash University) investigating cellular mediated allograft rejection following lung transplantation, focusing on both allo-specific and cross-reactive T cells. In 2014, she joined the Immunoproteomics Laboratory, headed by Prof Anthony Purcell (Monash University). She is an internationally recognised T cell immunologist with expertise in HLA immunogenetics, transplantation, cellular and viral immunology and T cell biology. Her laboratory examines T cell mediated investigations across different human disorders including transplantation, autoimmunity, allergy and cancer. Her research program is highly focused towards translational medicine in the pursuit of improved clinical outcomes for patients.
Our research
Current projects
1. Understanding the role of T cell cross-reactivity in human disorders
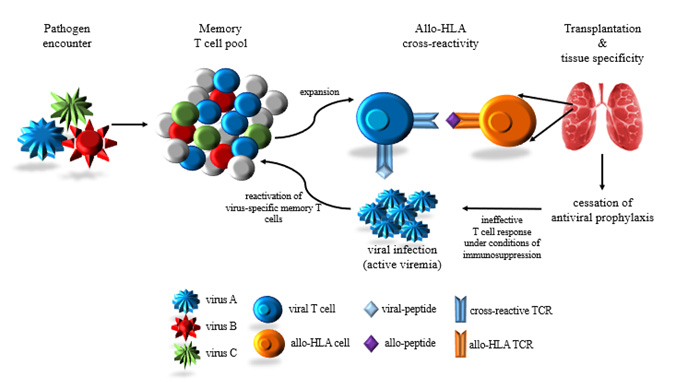
A hallmark of human antiviral T cells is their ability to recognise viral peptide antigen bound to a self-HLA on the surface of infected cells. Whilst this recognition often displays exquisite specificity, it is not uncommon for a subset of these T cells to cross-react on closely related peptide/HLA complexes. More remarkably, some T cells are also capable of recognising apparently distinct peptide/HLA complexes including non-self or allogeneic HLA (Mifsud et al., Am J Transplantation, 2008; Mifsud et al., Transplantation, 2010) and self-HLA expressed in different tissues. These forms of T cell cross-reactivity are not beneficial to the host and elicit inappropriate T cell responses that can lead to transplant rejection (reviewed in Rowntree et al., Curr Opin Organ Transplantation, 2016) and autoimmunity, respectively. These potentially hazardous T cell responses are the price paid to maintain immune potential to combat the vast array of pathogenic challenges encountered during a lifetime. We have identified cross-reactive virus-specific T cells (Nguyen et al., PLoS One, 2013; Nguyen et al., J Immunology, 2014) that require further functional characterisation to elucidate molecular interactions between the TCR/peptide/HLA complex and their potential contribution to adverse clinical events.
This project is in collaboration with A/Prof Tom Kotsimbos (Alfred Hospital), A/Prof Lloyd D’Orsogna (Fiona Stanley Hospital, Perth, WA), Prof Frans Claas (Leiden University Medical Centre, The Netherlands), Prof Linda Wooldridge (University of Bristol, UK).
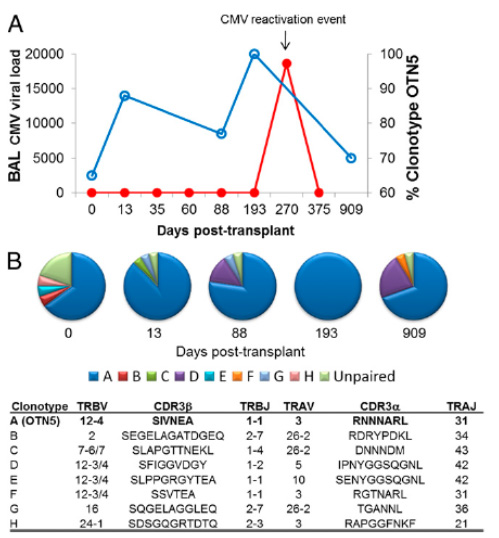
The figure above shows the cross-reactive OTN5 TCR (blue line, clonotype A) is detectable preceding, during and following a CMV reactivation episode in a lung transplant recipient (Nguyen et al., J Immunology, 2014).
2. T cell mediated cutaneous adverse drug reactions
Adverse drug reactions are one of the leading causes of death amongst hospitalised patients and create significant clinical management issues by restricting drug treatment options. Many drug reactions are T cell mediated and intimately linked with specific HLA allotypes, which has enabled characterisation of the underlying mechanisms (reviewed in Illing et al., Curr Opin Immunology, 2016). Importantly, this has led to worldwide regulatory recommendations for pre-treatment genetic screening for certain drug-HLA combinations (carbamazepine & HLA-B*15:02; abacavir & HLA-B*57:01) due to the 100% negative predictive value of such tests (i.e. no drug reaction if you do not express the HLA risk allele). However, the positive predictive value is much lower; 55% for abacavir hypersensitivity and only 5% for carbamazepine-induced severe skin reactions (reviewed in Mullan et al., HLA, 2019). Thus, a proportion of patients that may actually tolerate the treatment are precluded from using the drug. This indicates there are other immune-driven factors that contribute to the onset and clinical heterogeneity of adverse drug reactions. Our research has focused on the characterisation of drug-responsive T cells for abacavir, carbamazepine, allopurinol and beta-lactam antibiotics (i.e. penicillins), their specific recognition of self-peptide/HLA/drug complexes and other non-HLA-associated gene expression perturbations driving immunopathology.
This project is in collaboration with Prof Anthony Purcell and Dr Patricia Illing (BDI, Monash University), Prof Robyn O’Hehir and Prof Patrick Kwan (Alfred Hospital, RMH).
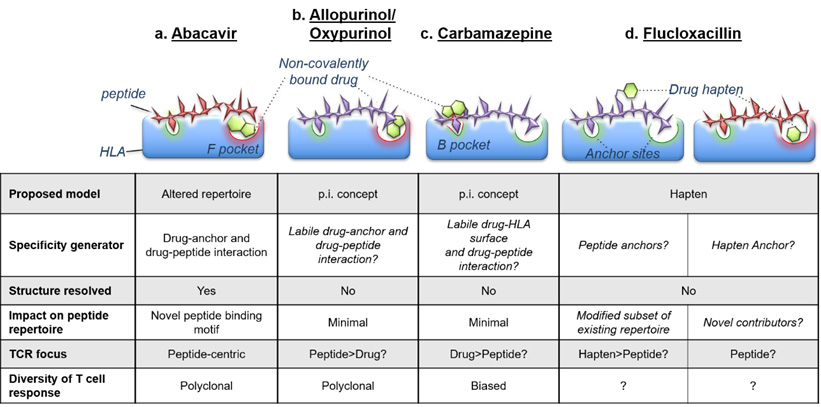
This figure shows the specificity of HLA-drug interactions is generated though interaction with HLA allotype-specific residues of the antigen-binding cleft (Illing et al., Curr Opin Immunology, 2016).
3. Identification of T cell epitopes for immunotherapeutic development
Our laboratory is actively involved in the identification of novel T cell targets for several human diseases, including cancer (e.g. haematopoietic malignancies, melanoma), autoimmunity (e.g. type I diabetes, ankylosing spondylitis) and infectious disease (e.g. influenza). These projects are designed to provide greater understanding of the functional and molecular triggers involved in disease immunopathology to facilitate disease prevention, development of prognostic tools and/or targeted therapeutics to improve patient outcomes.
These projects are in collaboration with Prof Anthony Purcell (BDI, Monash University), Prof Katherine Kedzierska (Peter Doherty Institute, University of Melbourne), A/Prof Alexandra Sharland (University of Sydney), Prof Anthony Schwarer (Box Hill Hospital).
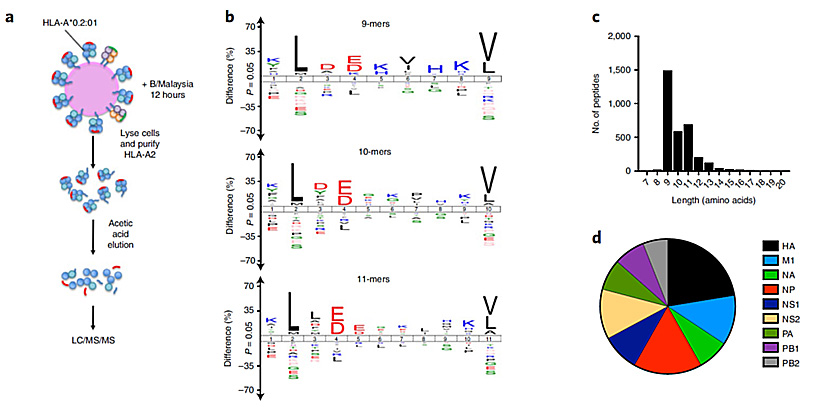
This figure shows the Identification of novel protective IBV CD8+ T cell epitopes by immunopeptidomics (Koutsakos et al., Nature Immunology, 2019).
Visit Dr Mifsud’s Monash research profile to see a full listing of current projects.
Research activities
My research expertise encompasses HLA immunology and immunogenetics, transplantation immunology, viral immunology and T cell biology. My main research focuses on how the antigen-specific T cell repertoire, shaped by previous exposure to pathogens, can influence future immune responses through TCR cross-reactivity. The intrinsically cross-reactive nature of T cells is greatly underappreciated and is important in the setting of destructive immunopathology and adverse clinical outcomes. These research findings will provide critical functional and structural information regarding the capacity of cross-reactive TCRs to initiate and/or contribute to common T cell mediated human diseases (e.g. autoimmunity, drug allergy and transplant rejection). The long-term clinical impact of my research has far-reaching potential given the fundamental nature of the immunology involved including translatable outcomes to aid clinicians in personalisation of patient clinical management, influence immunotherapeutic approaches, and alter drug design.
Key Research activities
- Transplantation immunology
- Viral immunology
- T cell cross-reactivity
- HLA immunogenetics
- Drug hypersensitivity
- Autoimmunity
- Antigen processing and presentation
Techniques/expertise
- Human T cells (lines and clones)
- Human DC cultures
- Human T cell receptor repertoire identification
- Transfection of cell lines
- Cellular immunoassays
- Generation of MHC/peptide class I tetramers
- Flow cytometry
- Protein expression and purification
- Molecular biology
- Proteomics (immunoaffinity chromatography, RP-HPLC and mass spectrometry)
Disease models
- Transplantation
- Haematopoietic malignancies and other cancers
- Drug hypersensitivity
- Autoimmunity
Genetically modified organisms
- Retrovirally transduced HLA class I molecules into various cell lines (C1R, 721.221, K562, T2)
- Retrovirally transduced TCRs into SKW3 cells
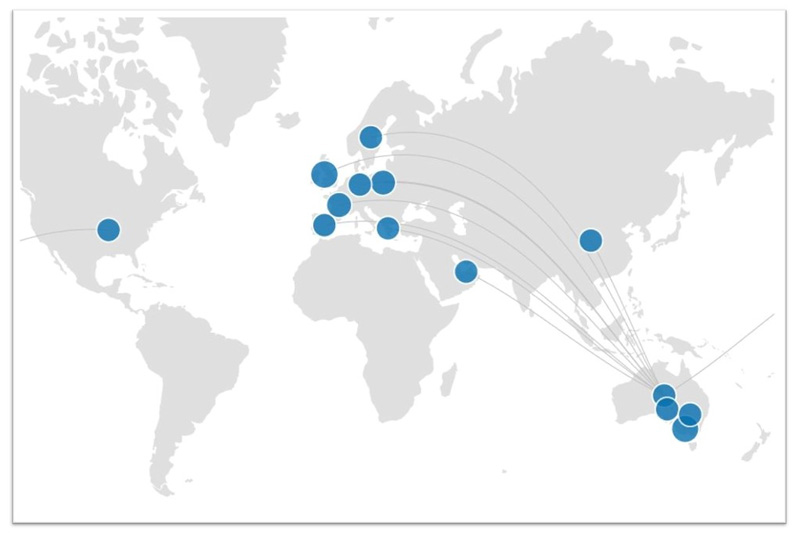
Collaborations
We collaborate with many scientists and research organisations around the world. Some of our more significant national and international collaborators are listed below. Click on the map to see the details for each of these collaborators (dive into specific publications and outputs by clicking on the dots).
Clinical:
A/Prof Tom Kotsimbos, Alfred Hospital
A/Prof Lloyd D'Orsogna, Fiona Stanley Hospital, WA
Prof John Wilson, Alfred Hospital
Prof Robyn O'Hehir, Alfred Hospital
Prof Patrick Kwan, Royal Melbourne Hospital & Alfred Hospital
Prof Anthony Schwarer, Box Hill Hospital
Dr George Grigoriadis, Monash Health and Translation Precinct
Professor Eric Morand, School of Clinical Sciences, Monash University
Scientific:
Prof Anthony Purcell, Monash University
Prof Jamie Rossjohn, Monash University
A/Prof Stephanie Gras, Monash University
A/Prof Lloyd D'Orsogna, Fiona Stanley Hospital, WA
Prof Linda Wooldridge, University of Bristol, England
Prof Mirjam Heemskirk, Leiden University Medical Centre, The Netherlands
Prof Frans Claas, Leiden University Medical Centre, The Netherlands
A/Prof Alexandra Sharland, University of Sydney, NSW
Prof Katherine Kedzierska, Peter Doherty Institute, University of Melbourne
Prof Weisan Chen, La Trobe University
Student research projects
The Mifsud Lab offers a variety of Honours, Masters and PhD projects for students interested in joining our group. There are also a number of short term research opportunities available.
Please visit Supervisor Connect to explore the projects currently available in our Lab.