Foldi Lab research
Collaborations | Student research projects | Publications
About Dr Claire Foldi
Dr Claire Foldi completed her PhD in Behavioural Neuroscience at the Queensland Brain Institute in 2012 under the supervision of Associate Professor Thomas Burne and Professor Darryl Eyles. Her doctoral thesis was focused on determining the biological plausibility of advanced paternal age as a risk factor for neurodevelopmental disorders, particularly schizophrenia and autistic spectrum disorders. Following Postdoctoral training in electrophysiology at the Karolinska Institutet in Stockholm, Sweden, she was recruited to the laboratory of Professor Brian Oldfield at Monash University in 2015. Dr Foldi now leads a program of research on anorexia and feeding disorders within the Metabolic Neurosciences Laboratory in the Department of Physiology.
Dr Foldi’s research priority is to understand the neurobiological mechanisms that underlie serious psychiatric disease, in particular anorexia nervosa and eating disorders. She is particularly interested in the often-unreported response heterogeneity that occurs in single neurons, animal models and patients in the clinic. She believes that interrogating the mechanisms underpinning this variability will inform the development of more effective treatment strategies.
Our research
Current projects
- The neurobiological causes of anorexia nervosa
- Mechanisms of novel pharmacotherapeutics for eating disorders
- Cognitive, behavioural and metabolic consequences of adolescent food insecurity
- Neural circuit and neurochemical drivers of compulsive exercise in activity-based anorexia
- Decoding the mechanisms of psychedelic-induced cognitive and neural plasticity
Visit Dr Claire Foldi's Monash research profile to see a full listing of current projects.
Research activities
1.Understanding the biological causes of anorexia nervosa, in order to find effective treatments
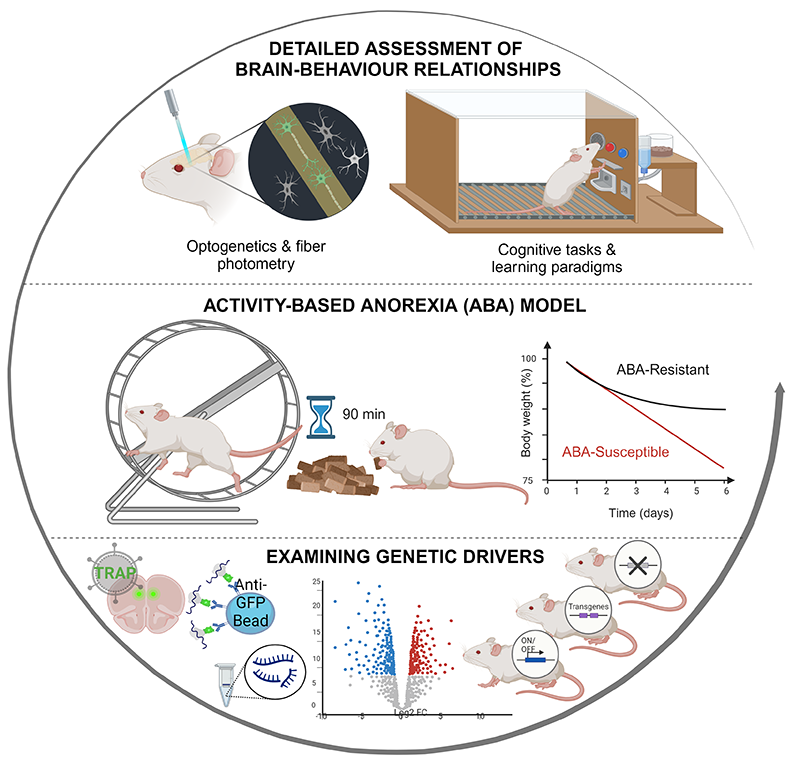
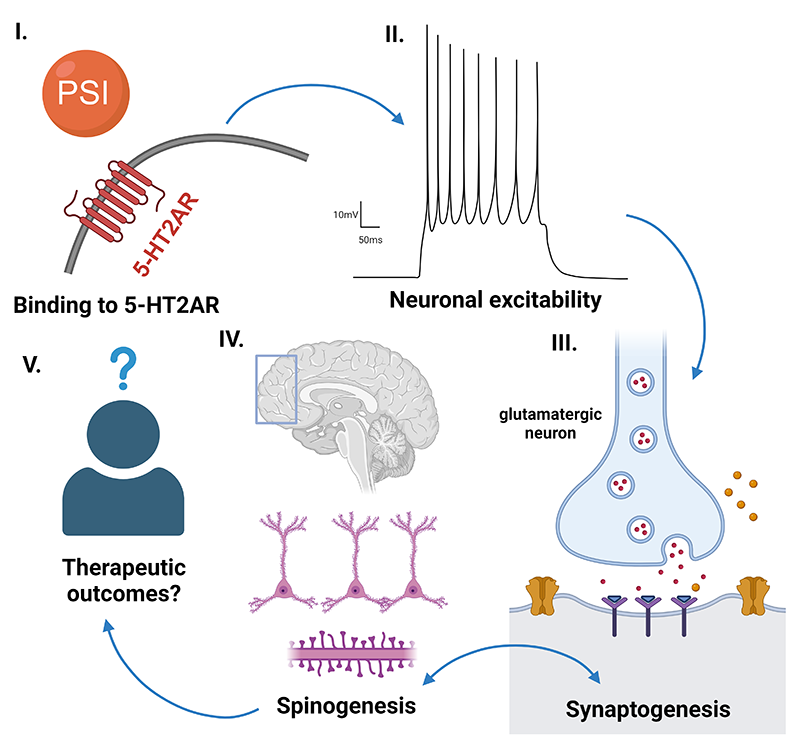
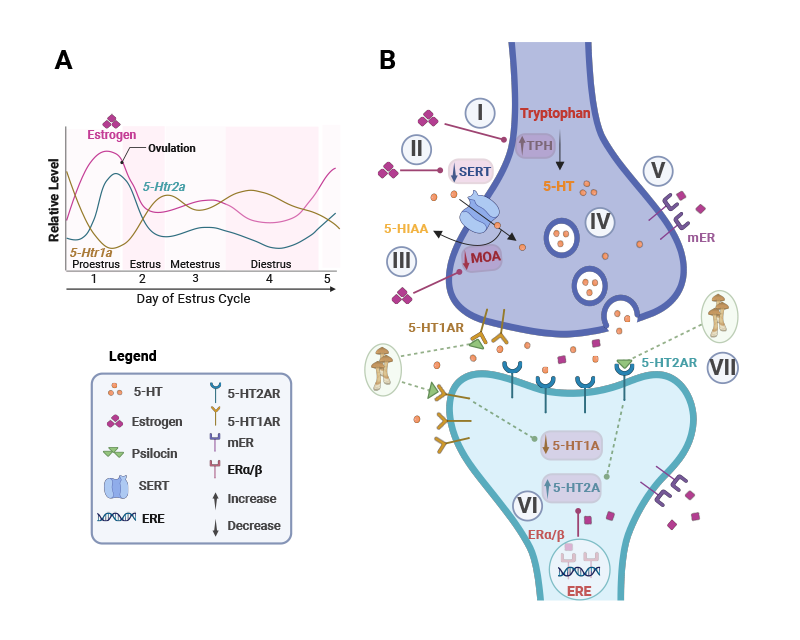
Our lab is the only research group in Australia working towards understanding the neural mechanisms underlying anorexia nervosa (AN), a debilitating mental health condition with the highest mortality rate of any mental disorder. As part of the Australian Eating Disorders Research and Translation Centre (AEDRTC), we are contributing to transforming the landscape of eating disorders research and translation. We use the activity-based anorexia (ABA) rat model to take brain targets identified in anorexia nervosa and dissect their causal role in the development of pathological feeding and exercise behaviour. One example is dysfunction in serotonin (5-HT) signalling in AN, that we have manipulated pharmacologically in ABA rats with the psychoactive compound psilocybin to examine the mechanisms through which psilocybin may be effective for treating AN.
TMM: https://pubmed.ncbi.nlm.nih.gov/38103992/
IJED: https://onlinelibrary.wiley.com/doi/10.1002/eat.24130
2.Exploring the effects of psychedelics on brain function and behaviour
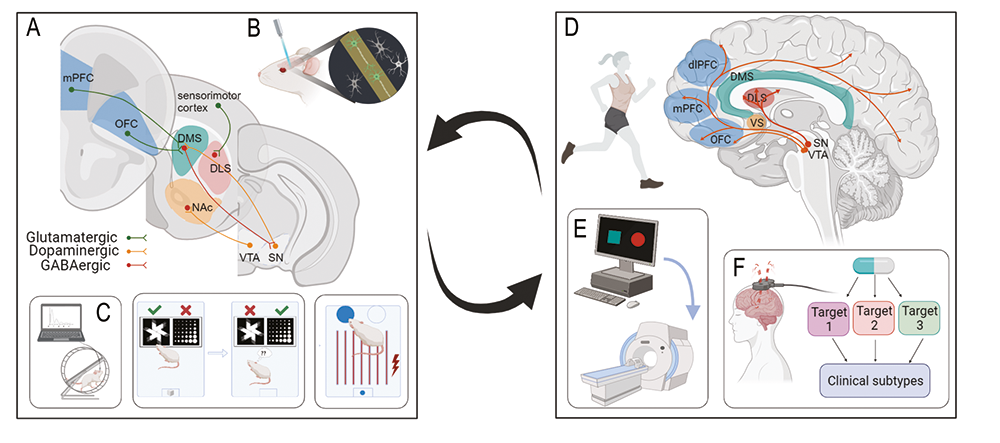
We are excited to uncover novel aspects of brain function influenced by psychedelics, not only to help inform their therapeutic application but also to gain insight into interacting neural circuits, and neurochemical dynamics. We are using optical recording approaches to reveal acute and enduring changes to brain function in response to psilocybin, and behavioural pharmacology techniques to understand how these changes relate to signalling through specific receptor mechanisms. The major behavioural changes we are investigating relate to decision-making, social motivation and food reward. We use two types of operant learning paradigms to assess cognitive behaviours, one that involves training animals to respond to visual stimuli on a touchscreen and one that involves training animals to nose-poke in specific sequences to obtain rewards. We are also measuring physiological effects of psychedelics on temperature regulation and gut-brain signalling, as well as whether specific aspects of psychedelic actions are influenced by sex hormones.
TIPS: https://www.sciencedirect.com/science/article/pii/S0165614723002766
Endo: https://academic.oup.com/endo/article/165/8/bqae083/7709979?searchresult=1
3. Defining the lasting consequences of adolescent food insecurity on brain function and mental health outcomes
We have also recently established a new line of research to determine how eating disorder symptoms arise from transient (adolescent) exposure to food insecurity, a major global health concern in the context of geopolitical instability and extreme weather events making nutritious food more difficult to reliably access. We see the metabolic, neurobiological and cognitive characterisation of adolescent exposure to food insecurity in both male and female mice as crucial to inform the development of public health mitigation strategies.
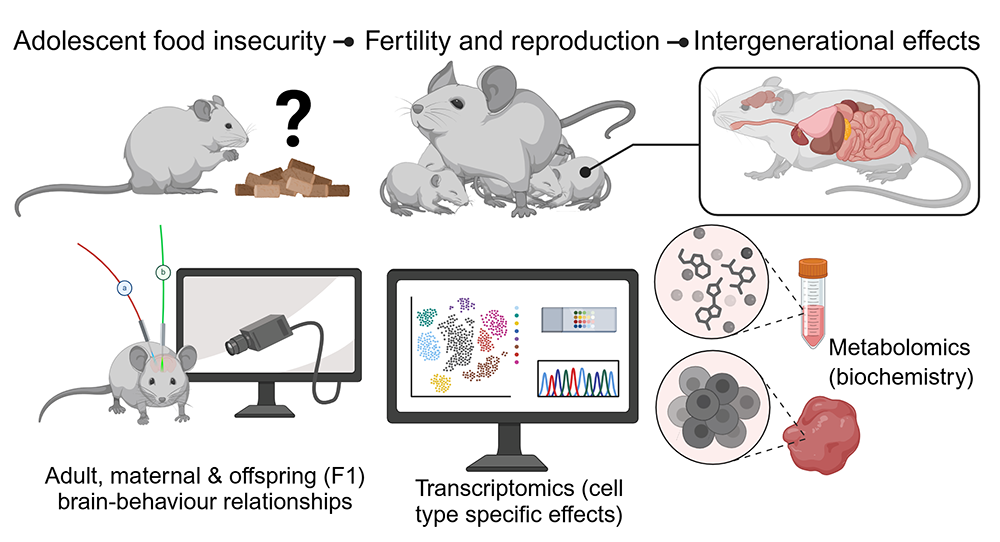
Transient food insecurity results in increased incidence of binge eating disorder, and we have developed a mouse model that supports a sex-dependent increase in binge eating symptoms. We are now examining whether there are intergenerational effects of adolescent food insecurity by screening offspring from mothers with a history of food insecurity.
Techniques/expertise
- Rat and mouse behaviour, learning and cognition
- Neural circuit tracing and manipulation
- Neural circuit function
- Behavioural pharmacology (focus on serotonin and dopamine)
- In vivo neuronal recording techniques (fiber photometry)
- Metabolic physiology
- Immunohistochemistry and imaging
- Gene and protein expression analyses
Disease models
The most well-accepted animal model of anorexia nervosa, known as activity-based anorexia (ABA), exploits the innate motivation of laboratory rats to run in wheels. When rats with access to running wheels are placed on a restricted feeding schedule, there is a paradoxical increase in running activity despite substantially decreased caloric intake, causing a profound reduction in body weight. It is important to recognise that food is only limited in terms of time and not quantity, and that animals quickly learn to increase their food intake to maintain body weight if this is the only parameter that is changed. Similarly, given access to running wheels and free access to food, they maintain their body weight well. It is only the combination of timed availability of food and access to running wheels that initiates the precipitous reduction in body weight that typifies the ABA model.
In the lab, we use the ABA model as a means to study factors that influence reward-based feeding, compulsive exercise behaviour and body weight maintenance.
Collaborations
Dr Foldi collaborates with an interdisciplinary team of researchers at Monash University, including Professor Zane Andrews (Physiology), Dr Paul Liknaitzky (Psychiatry), Professor Jakob Hohwy (Philosophy), Dr Sonika Tyagi (Alfred Health) and A/Prof Adeel Razi (Turner Institute).
Outside of Monash, Dr Foldi collaborates with A/Prof Lucy Palmer (Florey Institute for Mental Health Research), Drs Robyn Brown and Priya Sumithran (University of Melbourne), Dr Sasha Gorrell (University of California, San Francisco), Professor York Winter (Humboldt University, Berlin) and Professor Walter Kaye (University of California, San Diego).
“Dr Foldi is a member of both the Monash Centre for Consciousness and Contemplative Studies (M3CS) and the Monash Neuromedicines Discovery Centre (NDC). She also co-leads the Workforce Development stream of the National Eating Disorders Research and Translation Centre, a consortium led by the Inside Out Institute and University of Sydney and funded through a $13M federal grant.”
Click on the map to see the details for each of these collaborators (dive into specific publications and outputs by clicking on the dots).
Student research projects
The Foldi Lab offers a variety of Honours, Masters and PhD projects for students interested in joining our group. There are also a number of short term research opportunities available.
Please visit Supervisor Connect to explore the projects currently available in our Lab.

