Cancer Neural-Immune
Cancer diagnosis is a difficult journey that significantly impacts an individual's physical and emotional wellbeing.
Challenging circumstances can activate our body's fight-or-flight stress response. While this allows us to respond quickly in times of threat, if activated chronically, stress can have detrimental effects on our health.
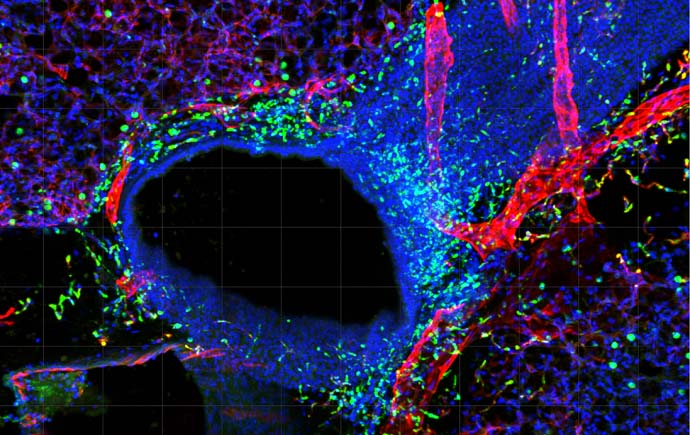
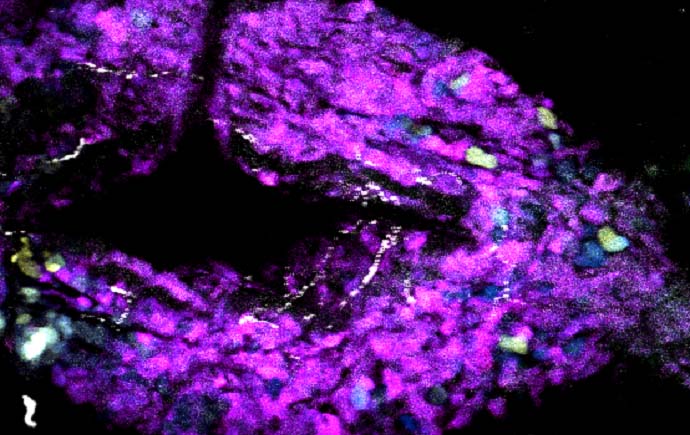
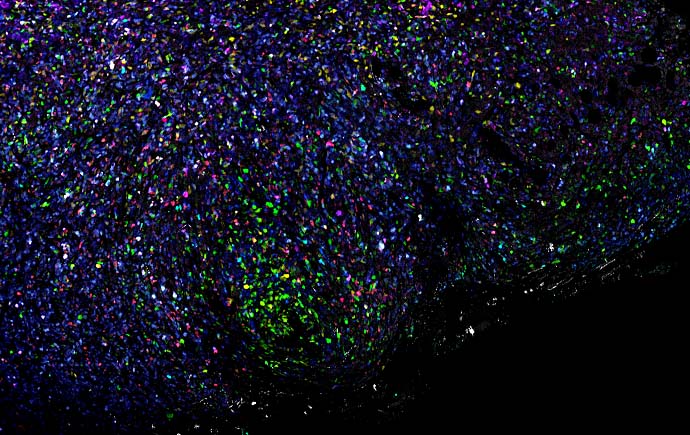
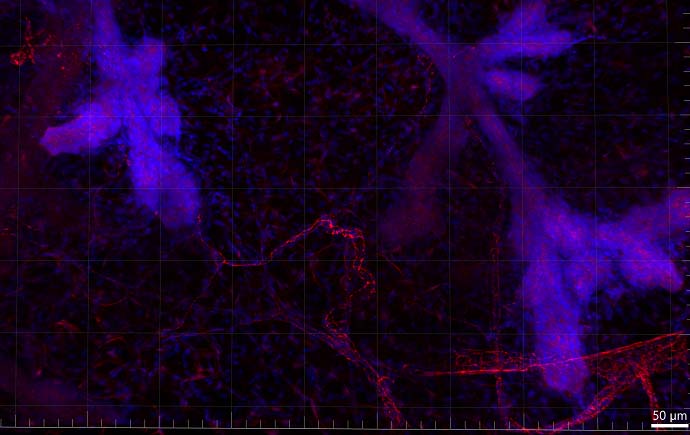
Our laboratory investigates the bi-directional interaction between the brain (or nervous system) and the tumour microenvironment. We use advanced imaging technology in animal models of cancer to investigate how stress signalling affects the progression of cancer and its response to therapy. Our research examines the impact of stress signalling that is initiated in the brain on cancer progression. We also investigate how the tumour talks to the brain to disrupt memory and learning, which in turn increases stress.
The goal of these studies is to identify the molecular and cellular impact of activating the sympathetic nervous system (SNS or fight-or-flight response) on cancer spread, and to understand the adverse side-effects of current treatments. These studies are helping us design better treatments to improve outcomes for cancer patients.
Group leaders
Learn more about us